Carbon Electron Configuration: If you guys have come across our recent article then it would be easy for you all to understand the concept. But if you are new here and looking for the information related to the carbon element and its electronic configuration, then today we will help you with some of the things and if you will be here till the last line surely you will go with some knowledge which can help you in your future and career.
- Oxygen Valency Electrons
- Bromine Electrons Configuration
- Blank Periodic Table Element
- Labelled Periodic Table
- Electronegativity Chart
- Lead Electron Configuration
- Vanadium Electron Configuration
- Nitrogen Electron Configuration
- Boron Electron Configuration
- Cesium Electron Configuration
- Strontium Electron Configuration
Carbon Electron Configuration
Chemistry is that subpart of science which is completely different from the other subjects and the students’ needs to be very attentive and smart to clear their doubts and if the students are just learning it for the sake of pass marks or any other reason then surely there will be a time when they will get stuck and will feel guilty that why they did not take the subject seriously.
You can easily practice it daily and get your concept clear. As today we are going to talk about an element that is very much complicated and there are many reasons given by many scientists but it is still difficult to predict which of the main answers is correct.
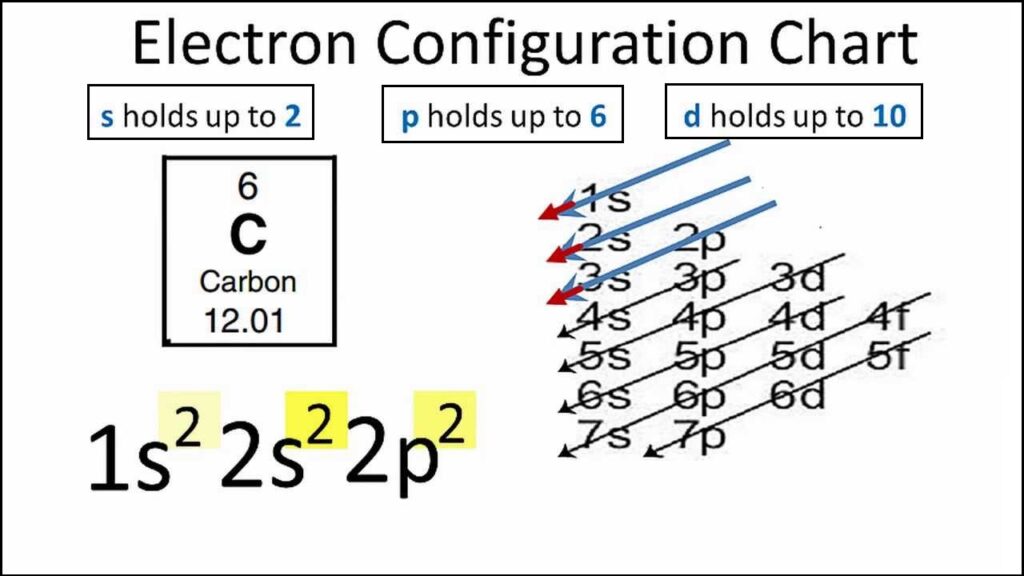
Carbon Electron Configuration is an element of the periodic table and its atomic number is 6. How many users might wonder what exactly will be the problem with that? The element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. The symbol of carbon is written as 6.
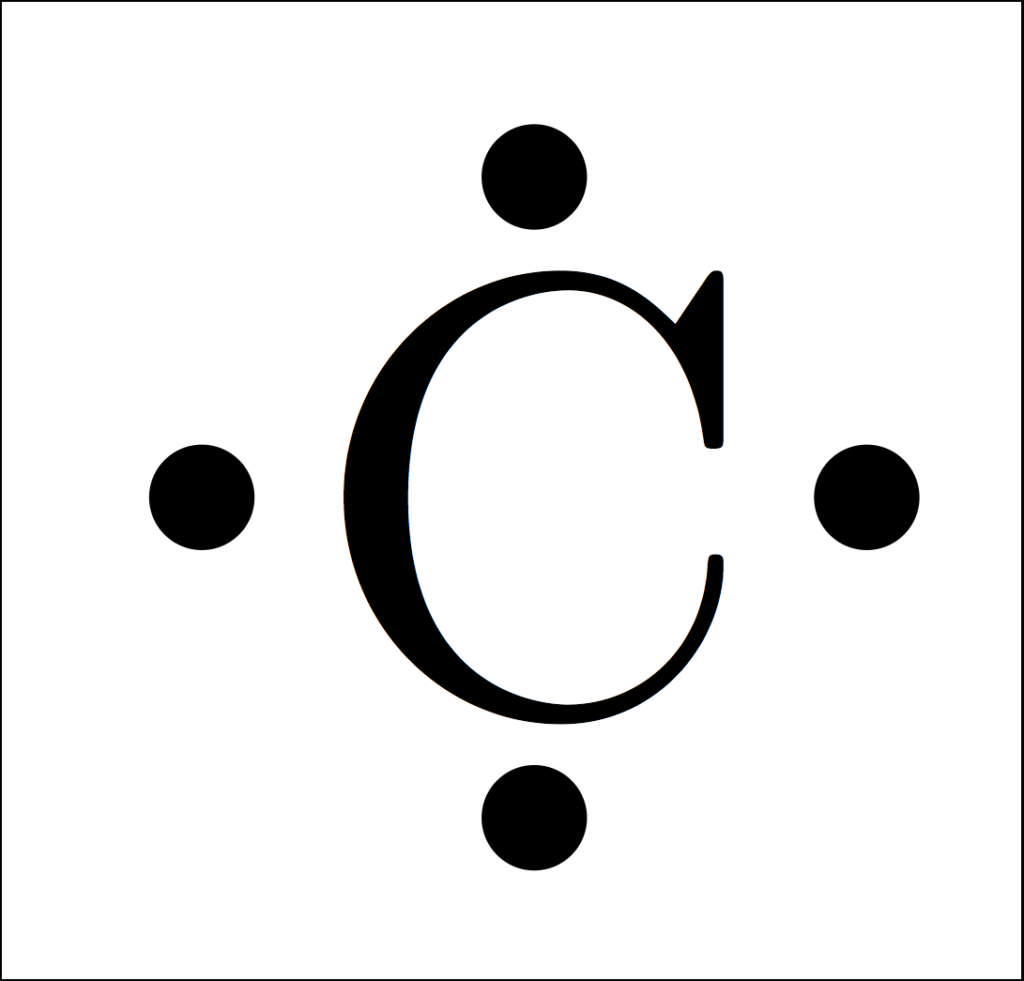
Carbon Electron Dot Diagram
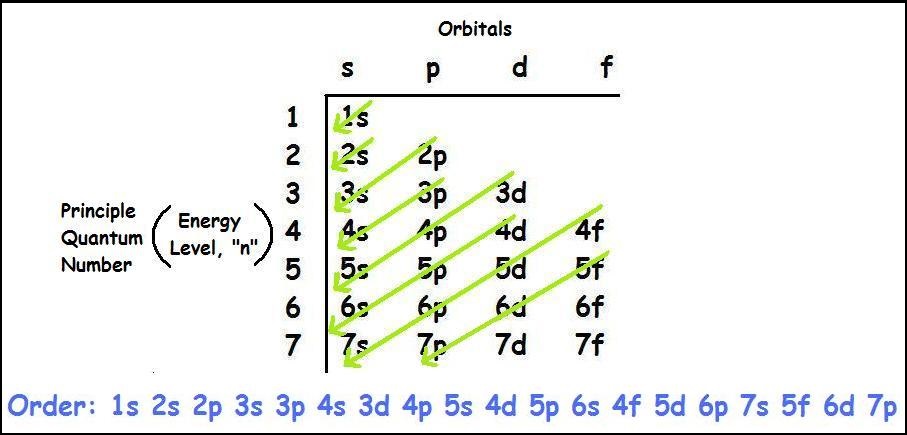
If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s22s22p2. Now the main thing here is what electronic configuration means, so the solution/ answer is that in simple words, by knowing the electronic configuration of any element we can know how many electrons are divided in their respective atomic orbitals.
It becomes easier to solve questions that are related to the finding of Carbon Electron Configuration. To help you with it, you can also view our image where you can get a clear vision of exactly how the electronic configuration of the element carbon is done. If we give you a brief about the details, then the first two electrons will be in the 1s orbit, and following the next will be two electrons in the 2nd orbit, and the ones which are left behind, will be counted in the 2p orbital.
Electron Configuration of Carbon
One major point before we come to the carbon electron dot diagram is that it is also called a Lewis dot diagram. Therefore the users don’t need to get confused between these two as both are the same. The reason behind the name given to Lewis is that it was firstly used by Gilbert N. Lewis.
Its main purpose is to tell the total number of Carbon Electron configurations (valence) in the atom, one more reason to use the dot diagram is that it provides us with the notations too. One good view of the diagram can be found here and you can get clear visibility most of you can refer here to get some of your concepts because knowing the diagram is also very important.
Carbon Electronic Configuration
As we have already provided you with the Carbon Electron Configuration element but it will be even easier to understand once you come to know its ways that which electron will go in which shell. For more information, you can download our sheet where you can get good knowledge regarding the electronic configuration along with all the elements.
The sheet is available free of cost and no charges are needed to use it. You can also save it in your storage devices such as your smartphone or laptop and whenever you need it you can visit any nearby stationery shop or printing shop and can get the hardcopy of it. You can use it for home purposes and school purposes too. It is open for all age groups.

Leave a Reply