Are you searching for the Periodic Table With Names, Atomic Mass and Number? The periodic table is also famous as the periodic table of elements. The elements in the table are analyzed in a manner so that anyone can easily know the properties of the elements.
The periodic table is necessary for all students, chemists as well as scientists also. Sometimes geologist also needs this to know the elements found on the surface. These tables include the information of elements like atomic mass, atomic number, electronegativity, ground-state level, atomic radius, etc. if you are working on particular properties, we also have a separate Periodic Table With Names And Atomic for every property. If you are a new learner or want to learn the table for the first time, you should download the basic periodic table.
We have a periodic table in the landscape as well as a portrait version. We also have tables in different sizes. We suggest you download the bigger one if you are going to print it out of the table. If you are comfortable with your device, you can also use the softcopy of the table to know the elements more.
Periodic Table of Elements with Names and Symbols PDF
Do you know there are many advantages of the Periodic Table of Elements with Names and Symbols PDF? In the periodic table, you will find the atomic number of the elements and some fundamental properties of the elements. It also helps to know about the elements in the group. It answers the most important questions about why elements show similar properties if they are in the same group and similarly why elements show different properties when they are in different groups.

It explains the properties of the elements and why it exists. It also explains why the properties of elements repeat after 2, 4, 8, and 32 elements. The periodic table helps us in easy learning as it will easy to learn the elements and their properties. It helps us to study our subject’s chemistry easily as well as systematically. Every element has its own space in the periodic table, which helps in easy learning.
Periodic Table With Names And Atomic Mass And Number
We should always learn something new to download the Periodic Table With Names And Atomic and know the elements or chemicals present on our earth. There are 118 chemical elements in the periodic table. If you learn the elements and their properties separately, then it won’t be very clear. So, scientists have made this wonderful periodic table to avoid confusion, and you can learn the periodic table easily. The elements are in the increasing form of the atomic number present in the periodic table.

Periodic Table with Full Names of Elements
Every element has some symbols, which are either one or two-digit. These special symbols help to know the elements easily. You will find the Periodic Table With Names And Atomic in every chemistry book, but do you know, our periodic table is so different.
You will find one or two tables in the books, but we have different periodic tables specially designed for your easy learning. Many people only need elements with atomic mass, elements with charge, elements with Atomic weight, etc. So, we have made the table accordingly. It will avoid your confusion, and you can focus on your aim with confidence.

Elements with Full Name with Atomic Number
If you are a scientist or science student, you can download the dynamic periodic table especially designed by the researchers. They have made this periodic table by giving the details of every property of the elements. If you have a dynamic periodic table, you will not need any other periodic table as it has all the information you need. We have the periodic table in printable format also.
So, you did not need to worry if you do not have a chemistry book, especially for the periodic table. You can take the printout of the Periodic Table With Names And Atomic in a4 sheet and can use it in your home, school, or laboratory. Carrying a single sheet of paper is not so tough, so you can easily use the periodic table according to your requirements.

Periodic Table with Names and Symbols
Only 94 elements present in the periodic are natural, and the rest are human-made. You will also know many more facts about the periodic table when you will download it. So, increase your knowledge of chemistry by downloading the amazing periodic table.
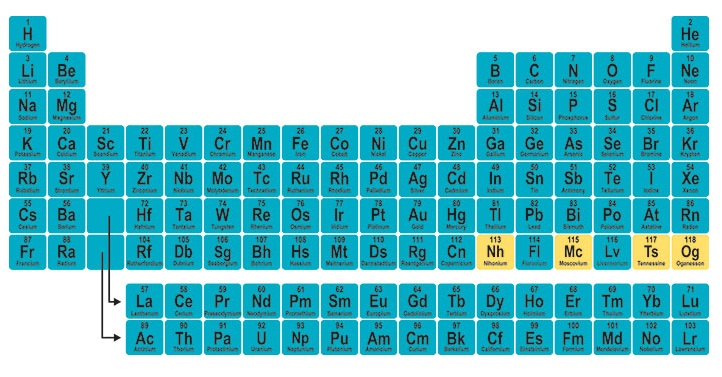
You can download Periodic Table with Names and Symbols by going on the downloading link. We have a periodic table in different colors also. So, you can get it according to your choice.
Leave a Reply